Technical Note : ข้อมูลทางเทคนิค
An Introduction to Transfection method
Life Science
Transfection describes the introduction of foreign material into eukaryotic cells. Transfection typically
describes the introduction of foreign material into eukaryotic cells. Transfection typically involves opening transient pores or holes in the cell plasma membrane, to allow the uptake of material. Genetic material (such as supercoiled plasmid DNA or siRNA constructs), or even proteins such as antibodies, may be transfected. Transfection is frequently carried out by mixing a cationic lipid with the material to produce liposomes, which fuse with the cell plasma membrane and deposit their cargo inside.
The term transfection is most often used in reference to mammalian cells, while the term transformation is used for the same process in bacteria and, occasionally, plants. The original meaning of transfection was infection by transformation, i.e. introduction of DNA (or RNA) from a eukaryote virus or bacteriophage into cells, resulting in an infection. Because the term transformation had another sense in eukaryotic cell biology (a genetic change allowing long-term propagation in culture, or acquisition of properties typical of cancer cells), the term transfection acquired, for animal cells, its present meaning of a change in cell properties caused by introduction of DNA.
Historical Background
The ability to introduce nucleic acids into cells has enabled the advancement of our knowledge of genetic regulation and protein function within eukaryotic cells, tissues and organisms. The successful pioneering studies of Vaheri and Pagano (1),
and Graham and van der Eb (2) with DEAEdextran and calcium phosphate-mediated transfection techniques, paved the way for future experiments necessitating DNA transfer into cultured eukaryotic cells. The process of introducing nucleic acids into cells by non-viral methods, such as the DEAE-dextran and calcium phosphate techniques, is defined as “transfection”. This process is distinct from “infection”, which is a viral method of nucleic acid introduction into cells.
Progress in transfection technology was relatively slow until the advent of molecular biology techniques for cloning plasmid DNA. These techniques provided the means to prepare and manipulate DNA sequences and the ability to prepare virtually unlimited amounts of relatively pure DNA for transfection experiments. Cloned sequences could also be used to generate RNA in vitrowith phage RNA polymerase using DNA templates with the corresponding polymerase promoter (3).
As the ability to prepare DNA and RNA for transfection became easier, additional methods, such as electroporation and liposome-mediated transfer, were developed to enable more efficient transfer of the nucleic acids to a broad range of cultured mammalian cells (4,5).
The development of reporter gene systems and selection methods for stable gene expression of transferred DNA greatly expanded the applications for gene transfer technology (Figure 1.1). In 1982, Gorman et al.initiated the reporter gene concept with the bacterial chloramphenicol acetyltransferase (CAT) gene and associated CAT assay system (6).
Using a reporter gene that is not endogenous to the cell, coupled with a sensitive assay system for that gene product, allows investigators to clone regulatory sequences of interest upstream of the reporter gene to study expression of the reporter gene under various conditions. This technology, together with the availability of transfection reagents, provides the foundation for studying promoter and enhancer sequences, trans-acting proteins such as transcription factors, mRNA processing, protein/ protein interactions, translation, and recombination events (7).
Since the introduction of the CAT gene and assay system several other reporter systems have been developed for various in vitroand in vivo applications including luciferase, -galactosidase, alkaline phosphatase and green fluorescent protein (7).
See Chapter 6 for detailed descriptions of Promega’s luciferase, CAT and -galactosidase reporter vectors and assay systems. Integration of DNA into the chromosome, or stable episomal maintenance, of reporter genes and other genes occurs with a relatively low frequency. The ability to select for these cells is made possible using genes that encode resistance to a lethal drug. An example of such a combination is the marker gene for neomycin phosphotransferase with the drug Geneticin® (8).
Individual cells that survive the drug treatment expand into clonal groups that can be individually selected, propagated and analyzed.
Today the study of gene regulation, the analysis of the expression and function of proteins within mammalian cells, the generation of transgenic organisms and in vivo/ex vivogene therapy strategies are all made possible by the availability of of gene transfer technologies, nucleic acid molecular biology and reporter gene systems.

Transfection Technologies
Many transfection techniques have been developed. Desirable features include high efficiency transfer of nucleic acid to the appropriate cellular organelle (for example, DNA into the nucleus), minimal intrusion or interference with normal cell physiology, low toxicity, ease of use, reproducibility, successful generation of stable transfectants, and in vivoefficacy. The techniques developed for gene transfer can be broadly classified as either chemical reagents or physical methods.
Chemical Reagents
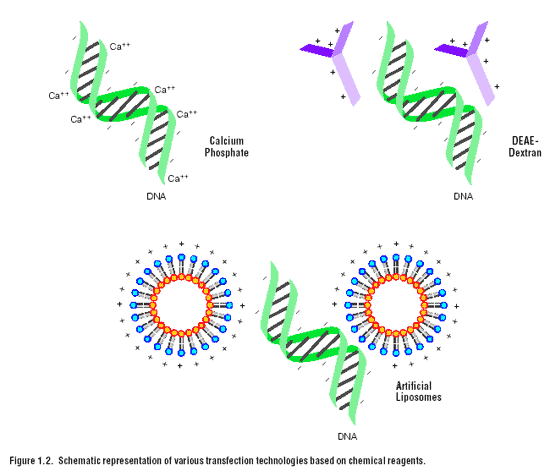
DEAE-dextran
was one of the first chemical reagents used for transfer of nucleic acids into cultured mammalian cells (1,9). The ProFectionฎ Mammalian Transfection System-DEAE-Dextran provides reagents for this transfection technique (see Chapter 4 for further information). DEAE-dextran is a cationic polymer that associates with negatively charged nucleic acids. An excess of positive charge, contributed by the polymer in the DNA/polymer complex allows the complex to come into closer association with the negatively charged cell membrane. Uptake of the complex is presumably by endocytosis. This method is successful for delivery of nucleic acids into cells for transient expression; that is, for short-term expression studies of a few days in duration. However, this technique is not generally useful for stable transfection studies that rely upon integration of the transferred DNA into the chromosome (10). Other synthetic cationic polymers have been used for the transfer of DNA into cells, including polybrene (11), polyethyleneimine (12) and dendrimers (13,14).
Calcium phosphate
co-precipitation became a popular transfection technique following the systematic examination of this method by Graham and van der Eb in the now-classic paper published in 1972 (2). Their study examined the effect of different cations, cationic and phosphate concentrations, and pH on the parameters of transfection. Calcium phosphate co-precipitation is widely used because the components are easily available and reasonable in price, the protocol is easy to use and many different types of cultured cells can be transfected. This method is routinely used for both transient and stable transfection of a variety of cell types. The protocol involves mixing DNA with calcium chloride, adding this in a controlled manner to a buffered saline/phosphate solution and allowing the mixture to incubate at room temperature. This step generates a precipitate that is dispersed onto the cultured cells. The precipitate is taken-up by the cells via endocytosis or phagocytosis. The calcium phosphate also appears to provide protection against intracellular and serum nucleases (15). Promega’s ProFection® Mammalian Transfection System-Calcium Phosphate provides reagents for this transfection technique (see Chapter 4 for further information).
By 1980, artificial liposomes were being used to deliver DNA into cells (5). The next advancement in liposomal vehicles was the development of synthetic cationic lipids by Felgner and colleagues (16). Liposome-mediated delivery offers advantages such as relatively high efficiency of gene transfer, ability to transfect certain cell types that are intransigent to calcium phosphate or DEAE-dextran, successful delivery of DNA of all sizes from oligonucleotides to yeast artificial chromosomes (16-20), delivery of RNA (21), and delivery of protein (22). Cells transfected by liposome techniques can be used for transient and for longer term experiments that rely upon integration of the DNA into the chromosome or episomal maintenance. Unlike the DEAE-dextran or calcium phosphate chemical methods, liposome-mediated nucleic acid delivery can be used for in vivotransfer of DNA and RNA to animals and humans (23).
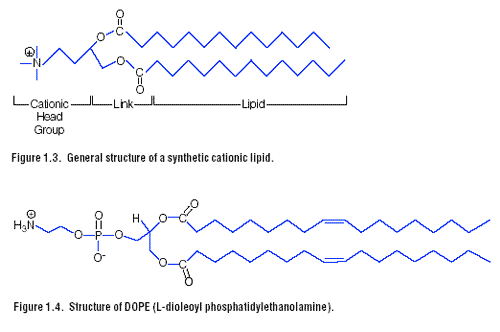
A lipid with overall net positive charge at physiological pH is the most common synthetic lipid component of liposomes developed for gene delivery (Figure 1.3). Often the cationic lipid is mixed with a neutral lipid such as L-dioleoyl phosphatidylethanolamine (DOPE) (Figure 1.4). The cationic portion of the lipid molecule associates with the negatively charged nucleic acids, resulting in compaction of the nucleic acid in a liposome/nucleic acid complex. For cultured cells, an overall net positive charge of the liposome/nucleic acid complex generally results in higher transfer efficiencies, presumably because this allows closer association of the complex with the negatively charged cell membrane. Following endocytosis, the complexes appear in the endosomes, and later in the nucleus. It is unclear how the nucleic acids are released from the endosomes and traverse the nuclear membrane. DOPE is considered a “fusogenic” lipid (24) and it is thought
 that its role may be to release these complexes from the endosomes, as well as to facilitate fusion of the outer cell membrane with the liposome/nucleic acid complexes. Promega provides a variety of transfection reagents that use cationic lipids for the delivery of nucleic acids to eukaryotic cells. These include TransFast Transfection Reagent, the Tfx Reagents and Transfectamฎ Reagent. See Chapter 3 for more information on the use of these reagents.
that its role may be to release these complexes from the endosomes, as well as to facilitate fusion of the outer cell membrane with the liposome/nucleic acid complexes. Promega provides a variety of transfection reagents that use cationic lipids for the delivery of nucleic acids to eukaryotic cells. These include TransFast Transfection Reagent, the Tfx Reagents and Transfectamฎ Reagent. See Chapter 3 for more information on the use of these reagents.
Physical Methods
Direct microinjection into cultured cells or nuclei is an effective, although laborious technique to deliver nucleic acids into cells. This method has been used to transfer DNA into embryonic stem cells that are used to produce transgenic organisms. However, this technique is not appropriate for studies that require a large number of transfected cells.
Electroporation
was first reported for gene transfer studies in 1982
This technique is often used for cell types such as plant protoplasts that are particularly recalcitrant to milder methods of gene transfer. The mechanism for entry into the cell is based upon perturbation of the cell membrane by an electrical pulse, which forms pores that allow the passage of nucleic acids into the cell.
The technique requires fine-tuning and optimization for duration and strength of the pulse for each type of cell used. A critical balance must be achieved between conditions that allow efficient delivery and conditions that kill cells.
Another physical method of gene delivery is biolistic particle delivery. This method relies upon high velocity delivery of nucleic acids on microprojectiles to recipient cells.
This method has been successfully employed to deliver nucleic acid to cultured cells, as well as to cells in vivo
Reference:
http://en.wikipedia.org/wiki/Transfection